(Nees ex Fr.) Greenhalgh & Morgan-Jones
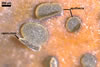
LEAF SPOTS at first single, small, 1-2 mm diam, later numerous, randomly distributed on the upper leaf surface, finally confluent and covering almost the whole upper leaf surface. In the laboratory, in moist chambers, first glistening swells, 0.6-1.0 mm diam, and later apothecia appeared on the spots.
 |
 |
 |
 |
 |
APOTHECIA sessile, slightly immersed in the tissue of the upper side of infected leaves, 0.6-1.0 mm diam, usually with upraised operculum attached to one of the lateral excipulum when mature.
Operculum consists of angular cells, 20-30 x 8-20 µm.
Excipulum consists of lateral and bottom excipulum.
 |
 |
 |
 |
 |
 |
 |
 |
 |
Lateral excipulum hyaline, maize yellow (4A6) to golden wheat (4B5), (16.5-)22.5-28.0(-27.9) µm thick, consisting of prolate, thin- to thick-walled cells, 2.0-6.6 µm wide, slightly broadened and rounded at their tops, usually forming feathery walls bended toward the outside of the apothecium.
Bottom excipulum yellowish brown (5D8) to brown (5E8), (13.2-)20.8-36.5(-42.6) µm thick, consisting of subcircular, angular, (3.2-)5.8(-9.3) µm diam, ovoid to irregular, 3.2-4.2 x 6.1-12.5 µm, thick-walled cells, tightly adherent to each other.
In PVLG |
In PVLG+Melzer's reagent |
||||
ASCI (a) clavate, straight or slightly curved, 6.6-9.6 x 57.6-93.4 µm, narrowed up to 2.5 µm at the base, with eight ascospores, and an apical pore, ca. 0.5 µm diam; asci separated by paraphyses.
Ascospores (as) ellipsoidal, hyaline, euseptate, 2.7-5.1 x 9.8-15.9 µm.
Paraphyses filiform, unbranched, hyaline, 1.8-2.2 µm and up to 4.2 µm wide at the base and the top, respectively, usually slightly longer than the asci.
In Melzer’s reagent, the apical pore of asci sometimes stained violet white (15A2), whereas ascospores always remained non-reactive.
DISTRIBUTION AND HABITAT. In Poland, The Western Pomerania province. On leaves of Ilex aquifolium L. growing in Szczecin, (53º26’N, 14º35’E), the Arboretum Glinna (53o18’N, 14o43’E), and the Arboretum Przelewice (53o06’N, 15o05’E) .
No other report exists of the occurrence of T. ilicina in Poland.
Literature data inform of records of T. ilicina in Germany (Rabenhorst 1896), Great Britain (Dennis 1968; Greenhalgh and Morgan-Jones 1964), temperate regions of USA (Farr et al. 1989), and Canada (Ginns 1986). According to G. Shannon (http://www.nifg.org.uk), this fungus was also found in many regions of Asia, Russia, southern Europe, and Africa.
NOTES. The apothecium of T. ilicina started to form under the cuticule, directly below the spots of the upper leaf surface. The first structure being formed was the bottom excipulum. Later, asci and paraphyses gradually rose from the bottom excipulum. When the development of the fungus proceeded, the fungal stroma increased the extent of its occurrence to the next layers of epidermis and slightly below it. In the last stage of development, ascospores originated. The developing apothecium inside the leaves, pressing on the under surface of the cuticule, first resulted in the origin of glistening swells on the leaf surface. Later, the swells burst and exposed the ascoma of the fungus. Freshly protruded apothecia were partly covered with the operculum that later detached.
The genus Trochila has been erected by Fries (1849). The first species of the genus was T. craterium (DC.) Fr. Other fungi included also were T. taxi Fr., R. ilicis (Schleicher) Fr., and T. laurocerasi (Desm.) Fr. In the same paper, Fries also listed Stegia ilicis Fr. Earlier, Chevalier (1826) transferred S. ilicis to the genus Eustegia Fr., and Rabenhorst (1844) to the genus Stegilla Reichb. Later, both Crouan and Crouan (1867) and Rehm (1896) transferred this fungus from Eustegia to Trochila as T. ilicis (Rabenh.) Crouan and T. ilicis (Chev.) Rehm, respectively. However, Dennis (1968) and Greenhalgh and Morgan-Jones (1964) concluded that the name T. ilicis (Chev.) Crouan used was a later homonym of T. ilicis (Schleicher) Fr. and, hence, invalid. Greenhalgh and Morgan-Jones (1964) examined specimens of Sphaeria ilicina Nees, a fungus mentioned by Fries (1823), and found it to be conspecific with T. ilicis (Chev.) Crouan. Consequently, they recombined this epithet as T. ilicina using the basionym S. ilicina Nees ex Fr.
The genus Trochila comprises ca. 15 fungal species. Of them, the only member of this genus found to colonize leaves of I. aquifolia is T. ilicina.
The morphological properties of T. ilicina encountered by the authors of this website generally fit those given by Rehm (1896), Greenhalgh and Morgan-Jones (1964), and Dennis (1968). Small differences found regard the size of asci and ascospores, as well as the reactivity of the apical pore of asci in Melzer’s reagent. The asci examined by us were slightly longer (up to 93.4 µm) than those earlier encountered (up to 80 µm; Rabenhorst 1896; Dennis 1968). Although the size of ascospores of the specimens examined by the authors of this website more or less overlapped with that cited in the literature, the ascospores of specimens coming from Poland were both slightly longer (up to 15.9 µm) and wider (up to 5.1 µm) than the longest (up to 12.5 µm; Greenhalgh and Morgan-Jones 1964) and the widest (up to 4.5 µm; Rabenhorst 1896; Greenhalgh and Morgan-Jones 1964; Dennis 1968) ascospores of specimens collected in other regions of Europe. Finally, all the authors mentioned above informed of amyloid properties of the apical pore of asci of T. ilicina, whereas the apical pore of asci of Polish specimens generally remained non-reactive or infrequently stained violet white (17A2) in Melzer’s reagent and a mixture of PVLG and Melzer’s reagent.
Although T. ilicina has been so far considered a saprotroph (Greenhalgh and Morgan-Jones 1964; Dennis 1968; Rehm 1896), the presence of spots on and the development of apothecia from leaves with no disease host symptoms tore off from their plant host suggest this fungus to be both a weak parasite and a saprotroph.
REFERENCES
Chevalier F. F. 1826. Flore général des environs de Paris. I. Paris.
Dennis R. W. G. 1968. British ascomycetes. 3301 Lehre Verlag Von J. Cramer.
Farr D. F., Bills G. F., Chamuris G. P., Rossman A. Y. 1989. Fungi on plants and plant products in the United States. APS Press. The American Phytopathological Society. St. Paul, Minnesota. USA.
Fries E. 1823. Systema Mycologicum 2.
Fries E. 1849. Summa Vegetabilum Scandinaviae.
Ginns J. H. 1986. Compendium of plant disease and decay fungi in Canada 1960-1980. Research Branch, Agriculture Canada Publication 1813.
Greenhalgh G. N., Morgan-Jones G. 1964. Some species of Trochila and an undescribed discomycete on leaves of Prunus laurocerasus. Trans. Brit. Mycol. Soc. 47: 311-320.
Rabenhorst L. 1844. Deutschlands Kryptogamen-Flora oder Handbuch zur Bestimmung der kryptogamischen Gewächse Deutschlands, der Schweiz, des Lombardisch-Venetianischen, Königreichs und Istriens, I. Leipzig.
Rehm H. 1896. Ascomyceten: Hysteriaceen und Discomyceten. In: Rabenhorst’s Kryptog.-Flora, Bd. 1, Abt. III.