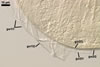
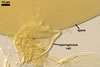
GERMINATION
SHIELD cardioid to irregular; pale yellow (2A3); 65-80 x 130-140
µm; formed on germinal wall 2.
AUXILIARY
CELLS rarely
single in the soil, usually in loose clusters of 2-5; hyaline
 |
In PVLG |
to pale yellow (4A3); lobed
to irregular; 18.0-22.5 x 27.5-32.5 µm; produced on coiled hyphae, 3.0-5.5
µm diam, concolourous with auxiliary cells.
MYCORRHIZAE.
Scutellospora nodosa has been associated in the field
with vesicular-arbuscular mycorrhizal roots of Salix triandra L.
(Blaszkowski 1991). Many attempts to initiate sporulation of this fungus in
both trap and one-species cultures failed.
PHYLOGENETIC POSITION.
According to De Souza et al. (2005), S. nodosa grouped within the clade A of the family Gigasporaceae between S. aurigloba and S. calospora.
DISTRIBUTION.
The
only known Polish habitat of occurrence of S. nodosa is that cited
in the original description of this fungus, i. e. under S. triandra
growing in a garden of Hel (54º36’N, 18º49’E) located in northern
Poland (Blaszkowski 1991).
NOTES. The unique morphological character of spores of S. nodosa is their knobby surface.
Scutellospora calospora, S. dipurpurescens, S. fulgida, and S. pellucida produce spores similar in size, shape, and colour to those of S. nodosa (Blaszkowski 1989; Franke and Morton 1994; Gerdemann and Trappe 1974; Koske and Walker 1986; Morton 1995, 2002; Morton and Koske 1988; Nicolson and Gerdemann 1968; Nicolson and Schenck 1979). However, the former four species have a smooth upper surface of their outermost spore wall layer that imparts a shiny appearance in reflected light under the dissecting microscope. In contrast, spores of S. nodosa are ornamented and, therefore, appear dull in reflected light.
Of the species listed above, the structure of the inner germinal walls of spores of S. nodosa most resembles that of spores of S. calospora and S. pellucida. In all these fungi, the germinal wall 1 is composed of two thin, flexible layers. The germinal wall 1 of S. dipurpurescens spores comprises only one layer. The next differences between these five species are in the number of inner germinal walls and the composition of the innermost germinal wall. Scutellospora fulgida differentiates only one germinal wall, lacking the germinal wall 2 of spores of S. nodosa. Two layers form the second germinal wall in spores of S. calospora, S. dipurpurescens and S. pellucida, whereas the germinal wall 2 of spores of S. nodosa consists of three layers.
As results from molecular analyses (De Souza et al. 2005), apart from S. calospora, the closest relative of S. nodosa also is S. aurigloba, a fungus producing small and much larger spores [323-428 x 323-469 µm vs. (160-)243(-270) µm diam when globose; Blaszkowski 1991; Walker and Hall 1991].
REFERENCES
Blaszkowski J. 1989.
Polish Endogonaceae. I. Acaulospora bireticulata, Entrophospora
infrequens, Glomus caledonium, and Scutellispora pellucida.
Karstenia 29, 1-10.
Blaszkowski J. 1991.
Polish Glomales 8. Scutellispora nodosa - a new species with knobby
spores. Mycologia 83, 537-542.
De Souza F. A., Declerck S., Smit E., Kowalchuk G. A. 2005. Morphological, ontogenetic and molecular characterization of Scutellospora reticulata (Glomeromycota). Mycol. Res. 109, 697-706.
Franke M., Morton J.
B. 1994. Ontogenetic comparisons of arbuscular mycorrhizal fungi Scutellospora
heterogama and Scutellospora pellucida: revision of taxonomic
character concepts, species descriptions, and phylogenetic hypotheses. Can.
J. Bot. 72, 122-134.
Gerdemann J. W., Trappe
J. M. 1974. The Endogonaceae in the Pacific Northwest. Myc. Memoir 5, 1-76.
Koske R. E., Walker
C. 1986. Species of Scutellospora (Endogonaceae) with smooth-walled
spores from maritime sand dunes: two new species and a redescription of the
spores of Scutellospora pellucida and Scutellospora calospora.
Mycotaxon 27, 219-235.
Morton J. B. 2002. International
Culture Collection of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi.
West Virginia University. http://www.invam.caf.wvu.edu/.
Morton J. M. 1995. Taxonomic and phylogenetic divergence among five Scutellospora species based on comparative developmental sequences. Mycologia 87, 127-137.
Morton J. B., Koske
R. E. 1988. Scutellospora dipurpurascens, a new species in the Endogonaceae
from West Virginia. Mycologia 80, 520-524.
Nicolson T. H., Gerdemann
J. W. 1968. Mycorrhizal Endogone species. Mycologia 60, 313-325.
Nicolson T. H., Schenck
N. C. 1979. Endogonaceous mycorrhizal endophytes in Florida. Mycologia 71,
178-198.