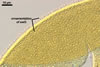
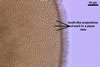
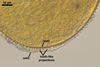
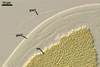
GERMINATION.
Not observed.
MYCORRHIZAE.
Many attempts to establish mycorrhizae of E. infrequens in one-species
cultures failed.
The only report of E. infrequens mycorrhizae established in a one-species culture probably is that of Sieverding and Toro (1986). According to these authors, E. infrequens formed typical vesicular-arbuscular mycorrhizae in roots of Pueraria phaseoloides Benth.
DISTRIBUTION.
Entrophospora infrequens has originally
been described as Gl. infrequens Hall from spores found in New Zealand.
Ames and Schneider (1979) concluded that Gl. infrequens was incompletely
described and based on spores wet sieved from a celery field soil of California
transferred this fungus to a newly established genus, Entrophospora
Ames & Schneider.
In Poland, E. infrequens
has been found in different both cultivated soils and those with natural
vegetation (Blaszkowski 1993a, b; Iwaniuk and Blaszkowski, unpubl.). However,
this fungus occurred rarely and in low abundances.
As literature data and
the observations of one of the authors of this website, J. Blaszkowski, indicate, E. infrequens
has a worldwide distribution. It has been encountered in, e. g., Canada
(Dalpé 1989), U. S. A. (Ames and Schneider 1979; Bloss and Walker 1987;
Halvorson and Koske 1987; Hetrick and Bloom 1983; Koske and Halvorson 1989;
Pfleger and Steward 1989; Schenck and Smith 1982; Stahl and Christensen 1982),
France, Switzerland and Germany (Oehl et al. 2003), Turkey (Blaszkowski, pers.
observ.), India (Sridhar and Beena 2001), New Zealand (Hall 1977), and Australia
(Hall and Abbott 1984).
NOTES.
Entrophospora infrequens is the type of the genus Entrophospora
that includes four species at present. Another species of this genus forming
spores with a structural wall layer ornamented with outgrowths is E.
baltica Blaszk. However, the ornamentation of the latter species
consists of small warts (Blaszkowski et al. 1998) rather than tooth-shaped
outgrowths as in E. infrequens (Ames and Schneider 1979; Blaszkowski
1989). Additionally, spores of E. baltica are surrounded with a hyphal
mantle that is lacking in spores of E. infrequens.
REFERENCES
Ames R. N., Schneider
R. W. 1979. Entrophospora, a new genus in the Endogonaceae. Mycotaxon
8, 347-352.
Blaszkowski J. 1989.
Polish Endogonaceae. I. Acaulospora bireticulata, Entrophospora
infrequens, Glomus caledonium, and Scutellispora pellucida.
Karstenia 29, 1-10.
Blaszkowski J. 1993a.
The occurrence of arbuscular fungi and mycorrhizae (Glomales) in plant communities
of maritime dunes and shores of Poland. Bull. Pol. Ac. Sci. Biol. Sci. 41,
377-392.
Blaszkowski J. 1993b.
Comparative studies of the occurrence of arbuscular fungi and mycorrhizae
(Glomales) in cultivated and uncultivated soils of Poland. Acta Mycol. 28,
93-140.
Blaszkowski J., Madej
T., Tadych M. 1998. Entrophospora baltica sp. nov. and Glomus
fuegianum, two species in the Glomales from Poland. Mycotaxon 68, 165-184.
Bloss H. E., Walker
C. 1987. Some endogonaceous mycorrhizal fungi of the Santa Catalina Mountains
in Arizona. Mycologia 79, 649-654.
Dalpé Y. 1989.
Inventaire et repartition de la flore endomycorhizienne de dunes et de rivages
maritimes du Quebec, du Nouveau-Brunswick et de la Nouvelle-Ecosse. Naturaliste
can. (Rev. Ecol. Syst.) 116, 219-236.
Hall I. R. 1977. Species
and mycorrhizal infections of New Zealand Endogonaceae. Trans. Br. Mycol.
Soc. 68, 341-356.
Hall I. R., Abbott L.
K. 1984. Some Endogonaceae from south western Australia. Trans. Br. Mycol.
Soc. 83, 203-208.
Halvorson W. L., Koske
R. E. 1987. Mycorrhizae associated with an invasion of Erechtites glomerata
(Asteraceae) on San Miguel Island, California. Madrono 34, 260-268.
Hetrick B. A. D., Bloom
J. 1983. Vesicular-arbuscular mycorrhizal fungi associated with native tall
grass prairie and cultivated winter wheat. Can. J. Bot. 61, 2140-2146.
Koske R. E., Halvorson
W. L. 1989. Mycorrhizal associations of selected plant species from San Miguel
Island, Channel Islands National Park, California. Pacific Sci. 43, 32-40.
Oehl F., Sieverding E.,
Ineichen K., Mader P., Boller T., Wiemken A. 2003. Impact of land use intensity
on the species diversity of arbuscular mycorrhizal fungi in agroecosystems
of Central Europe. Appl. Environ. Microbiol.69, 2816-2824.
Pfleger F. L., Steward
E. L. 1989. Survey of the Endogonaceae in Minnesota with synoptic keys to
genera and species. J. Minnesota Ac. Sci. 54, 25-29.
Schenck N. C., Smith
G. S. 1982. Additional new and unreported species of mycorrhizal fungi (Endogonaceae)
from Florida. Mycologia 74, 77-92.
Sieverding E., S. Toro T. 1986. The genus Entrophospora in Colombia. In: Gianinazzi-Pearson V., Gianinazzi S. (eds). Physiological and genetical aspects of mycorrhizae. Proc. 1st European Symposioum on Mycorrhizae. Dijon, 1-5 July 1985, 621-626.
Sridhar K. R., Beena
K. R. 2001. Arbuscular mycorrhizal research in coastal sand dunes: a review.
Proc. Nat. Acad. Sci. India. 71, 179-205.
Stahl P. D., Christensen
M. 1982. Mycorrhizal fungi associated with Bouteloua and Agropyron
in Wyoming sagebrush-grasslands. Mycologia 74, 877-885.