Glomus
mosseae
(Nicol.
& Gerd.) Gerd. & Trappe
SPORES
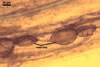
single in the soil, in loose aggregates or compact sporocarps;
pale yellow (2A2) to golden yellow (5B8); globose to subglobose; (80-)185(-280)
µm diam; sometimes irregular; 80-140 x 195-280 µm; with one subtending
hypha. Sporocarps
contain 2-5 spores surrounded with a peridium. Peridium composed of loosely
interwoven, branched, hyaline hyphae, 1.5-16.0 µm wide, with walls 0.5-0.8
µm thick.
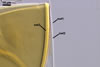
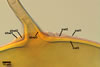
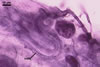
SUBCELLULAR
STRUCTURE OF SPORES consists of one wall with three layers
(swl1-3).
|
|
|
|
|
|
|
PVLG |
In PVLG+Melzer's reagent
|
|
|
|
|
|
|
|
In PVLG+Melzer's reagent |
Layer
1 mucilagenous, hyaline, (0.5-)1.1(-2.0) µm thick,
staining reddish white (11A2) in Melzer’s reagent, usually present
only in the most juvenile spores.
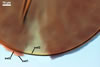
Layer
2 semiflexible, smooth, hyaline, (0.8-)1.2(-1.8) µm
thick, rarely present in mature spores, usually visible in the form of highly
decomposed fragments.
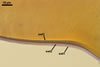
Layer
3 laminate, pale yellow (2A2) to golden yellow (5B8), (2.8-)4.5(-7.2)
µm thick.
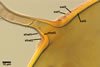
GERMINATION.
A
germ tube emerges from the lumen of the subtending hypha.
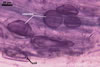
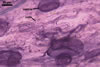
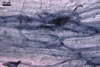
MYCORRHIZAE.
In roots of Plantago lanceolata L., mycorrhizae of Gl.
mosseae consisted of arbuscules, vesicles, as well as intra- and extraradical
hyphae staining intensively in 0.1% trypan blue.
|
|
|
|
|
|
|
|
|
|
|
|
In roots of P. lanceolata
|
DISTRIBUTION.
Glomus mosseae
is a frequent component of communities of arbuscular mycorrhizal fungi associated
with plants of different regions of the world (Blaszkowski 1993; Blaszkowski
et al. 2001). Blaszkowski (1993) found it to be the third most frequently
occurring arbuscular fungal species in Poland; it markedly preferred cultivated
soils. In uncultivated Polish sites, Gl. mosseae has been revealed
in the Bledowska Desert (50o22’N, 19o34’E; Blaszkowski et al.
2002), the Hel Peninsula (54o47’N, 18o25’E-54o36’N, 18o49’E;
Blaszkowski 1994), and the Tuchola Forests (53o46’N, 17o42’E-5340’N,
17o54’E; Tadych and Blaszkowski 2000).
NOTES.
When observed under a dissecting microscope, spores of Gl.
mosseae are most similar to those of Gl.
caledonium. However, the spore wall of the former species contains
three layers, of which two outer ones slough with age The latter fungus produces
spores with a 4-layered wall and the impermanent layer is only the outermost
one (Morton 1996, 2000).
REFERENCES
Blaszkowski J. 1993.
Comparative studies of the occurrence of arbuscular fungi and mycorrhizae
(Glomales) in cultivated and uncultivated soils of Poland. Acta Mycol. 28,
93-140.
Blaszkowski J. 1994.
Arbuscular fungi and mycorrhizae (Glomales) of the Hel Peninsula, Poland.
Mycorrhiza 5, 71-88.
Blaszkowski J., Tadych
M., Madej T., Adamska I., Iwaniuk A. 2001. Arbuscular mycorrhizal fungi (Glomales,
Zygomycota) of Israeli soils. Mat. II Polsko-Izraelskiej Konf. Nauk. nt. „Gospodarowanie
zasobami wodnymi i nawadnianie roslin uprawnych”. Przeglad naukowy Wydz.
Inz. Ksztalt. Srod. 22, 8-27.
Blaszkowski J., Tadych
M., Madej T. 2002. Arbuscular mycorrhizal fungi (Glomales, Zygomycota) of
the Bledowska Desert, Poland. Acta Soc. Bot. Pol. 71, 71-85.
Morton J. M. 1996. Redescription
of Glomus caledonium based on correspondence of spore morphological
characters in type specimens and a living reference culture. Mycorrhiza 6,
161-166.
Morton J. B. 2000. International
Culture Collection of Arbuscular and Vesicular-Arbuscular Mycorrhizal Fungi.
West Virginia University.
Tadych M., Blaszkowski
J. 2000. Arbuscular mycorrhizal fungi of the Brda river valley in the Tuchola
Forests. Acta Mycol. 35, 3-23.